$106.00
Beautiful full-color photography, effective illustrations, carefully written exercises, and a reasonable price combine to make Microbiology: Laboratory Theory & Application the best-selling microbiology lab manual series on the market. This edition satisfies the content needs of courses populated by majors or in nonmajors courses where an increased coverage of applied microbiology is desired.
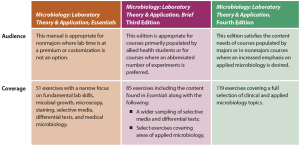
The following grid illustrates the primary audience and coverage differences between the three titles in the Microbiology: Laboratory Theory and Application series.
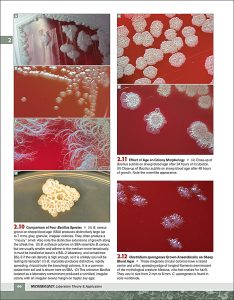
Examples of interior pages:

Instructor Resources Available: Login to view Instructor Resources